 
Explain the important aspects of resonance with reference to the C0 3 2- ion. Bond-lengths are measured by spectroscopic methods. 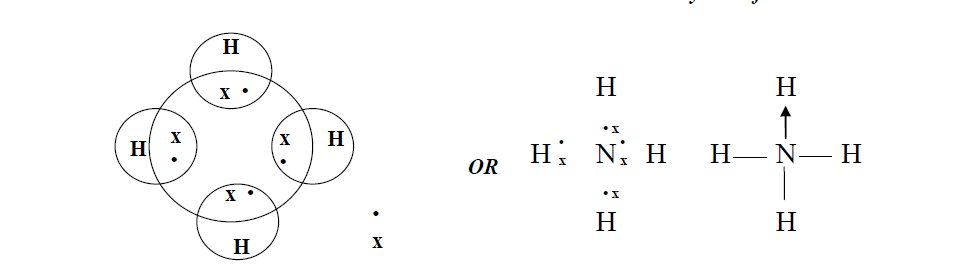
Greater the bond order, more is the bond strength.Īnswer: Bond-length: It is the equilibrium distance between the nuclei of two bonded atoms in a molecule. How do you express the bond strength in terms of bond order?Īnswer:Bond strength is directly proportional to the bond order. Thus, the bond angle is less in H 20 molecules. Discuss.īecause of two lone pairs of electrons on O-atom, repulsion on bond pairs is greater in H 20 in comparison to NH 3 . Although geometries of NH 3 and H 20 molecules are distorted tetrahedral, bond angle in water is less than that of ammonia. Discuss the shape of the following molecules using the VSEPR model:īeCl 2, BCl 3, SiCl 4, AsF 5, H 2S, PH 3 High lattice enthalpy of compound formed.High electron gain enthalpy of non-metal atoms.Write the favourable factors for the formation of ionic bond. This theory does not account for shape of molecules.Therefore, validity of the octet rule has been challenged. However, some of noble gas elements like Xenon have formed compounds with fluorine and oxygen. According to Octet rule, atoms take part in chemical combination to achieve the configuration of nearest noble gas elements.Significance: It helps to explain why different atoms combine with each other to form ionic compounds or covalent compounds. Write its significance and limitations.Īnswer: Octet rule: Atoms of elements combine with each other in order to complete their respective octets so as to acquire the stable gas configuration. Draw the Lewis structures for the following molecules and ions: Write Lewis symbols for the following atoms and ions: S and S 2– Al and Al 3+ H and H – Question 2.Write Lewis dot symbols for atoms of the following elements: Mg, Na, B, O, N, Br. This can occur in two ways by transfer of one or more electrons from one atom to other or by sharing of electrons between two or more atoms. Explain the formation of a chemical bond.Īnswer: According to Kossel and Lewis, atoms combine together in order to complete their respective octets so as to acquire the stable inert gas configuration. NCERT Solutions Class 11 Chemistry Chemistry Lab Manual Chemistry Sample Papers NCERT TEXTBOOK QUESTIONS SOLVED The Valence Shell Election Pair Repulsion (VSEPR) Theoryīonding in Some Homonuclear Diatomic Molecules Kössel-Lewis Approach to Chemical Bonding Topics and Subtopics in NCERT Solutions for Class 11 Chemistry Chapter 4 Chemical Bonding and Molecular Structure : Section Name 
NCERT Solutions for Class 11 Chemistry Chapter 4 Chemical Bonding and Molecular Structure
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
